Michael Howerton, UC San Francisco
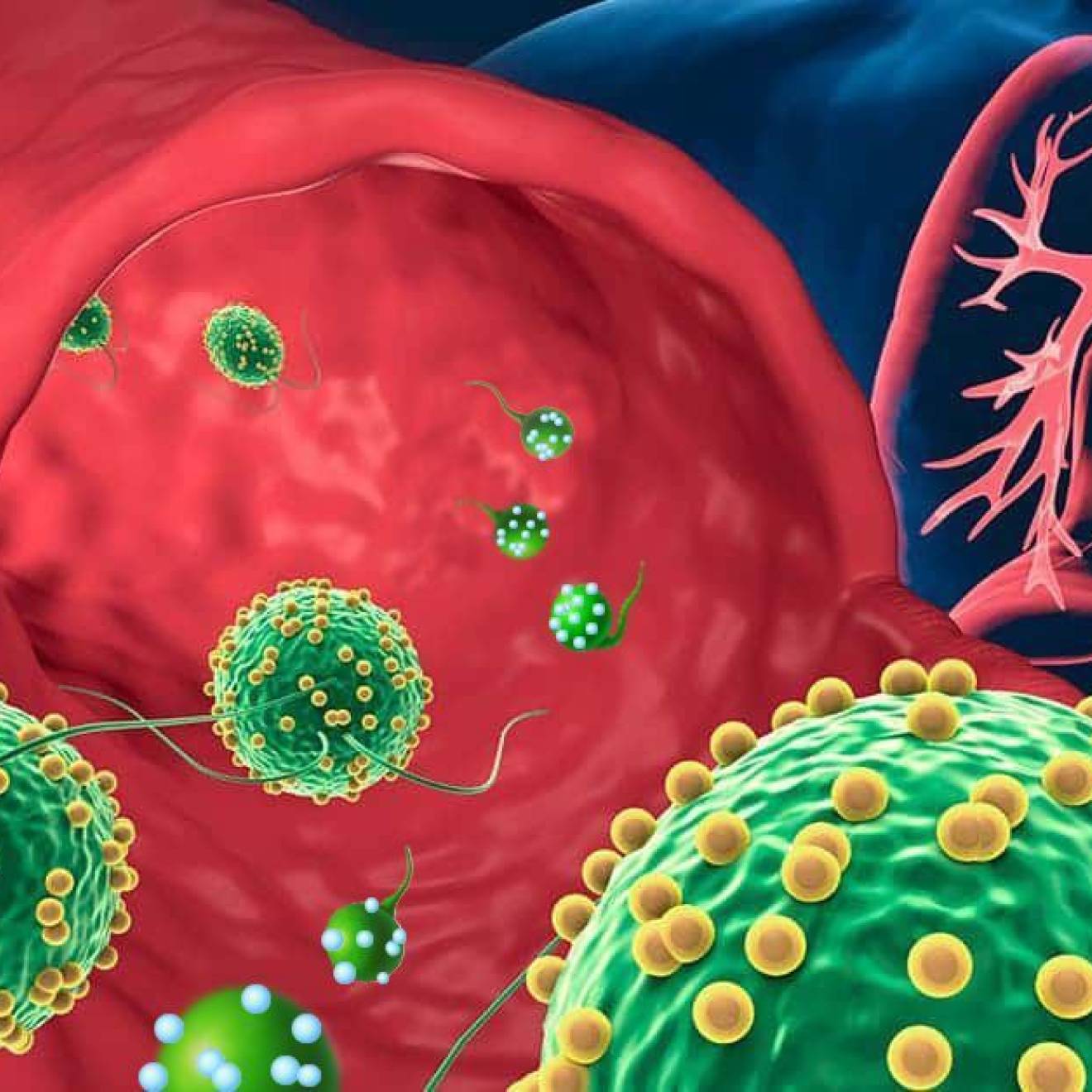
One of the confounding characteristics of cancer has long been that the body’s usually active patrol against viruses tends to leave deadly cancer cells alone to fester, mutate and spread.
The immune system has this blind spot by design — an immune system that has an ability to attack itself leads to autoimmune diseases, so as protection, it screens out its own tissue.
For decades, scientists assumed that cancer was beyond the reach of the body’s natural defenses. But after decades of skepticism that the immune system could be trained to root out and eliminate these malignant cells, a new generation of drugs is proving otherwise.
The treatment consists of infusing antibodies that enhance the immune system to recognize cancer cells and attack it. What’s more, since the immune system has a built-in memory, it continues to go after cancer cells, so the response can be longer lasting and more complete.
The trick is that this treatment doesn’t work for everybody, and researchers don’t yet understand why. But when it does work, the results have been particularly impressive.
“Although there is a 30-year history of people and institutions trying to develop immunotherapy approaches to cancer, it has only been in the last 10 years that we’ve broken through and have been able to impact cancer using immunotherapy,” said Jeffrey Bluestone, Ph.D., executive vice chancellor and provost of UC San Francisco.
“I do think that we’re at an inflection point with immunotherapy,” he added. “It will be revolutionary and will impact how we approach cancer for years to come.”
Treating the patient instead of the disease
"For the longest time, people did not believe this was possible,” said Lawrence Fong, M.D., associate professor of medicine at UCSF and one of the University’s lead investigators in the expanding use of immunotherapy.

Credit: Susan Merrell
“Now we can treat cancer by treating the patient instead of the disease,” he said. “That’s the biggest change. We can treat cancer without delivering chemotherapy or radiation to kill the cancer or performing surgery to get rid of the tumor.”
Researchers at UCSF and elsewhere have identified cell receptors, such as CTLA-4 and PD-1, which act as a brake on the immune system, limiting its response.
With the use of antibodies to inhibit these blockade receptors, allowing a more active and vigilant immune system, doctors have seen outstanding responses in patients with metastatic melanoma and lung cancer, both of which are almost always fatal with conventional treatments. Immunotherapy also has been successful in cancers of the bladder, prostate, kidney and bone marrow.
“This was a radical idea: that the body already possesses the ability to defeat cancer, and that medicine’s role was to find a way to allow the body to marshal the healing work it is naturally capable of,” Fong said. “We all believed it could work, but very few would have predicted the 180-degree change that we’ve seen over the last two years.”
Ever since James Allison, Ph.D., chair of the University of Texas MD Anderson Cancer Center’s Immunology Department, developed the first drug that increased survival for patients with metastatic melanoma – an antibody against CTLA-4 that became ipilimumab and was approved by the U.S. Food and Drug Administration in 2011 — it has been like a door has swung open.
In December, Allison won one of the 2014 Breakthrough Prize in Life Sciences for his groundbreaking discovery.
Indeed, the treatment has been a hot topic at recent cancer conferences and all top pharmaceutical companies are developing immunotherapy drug programs. Science magazine declared immunotherapy its top breakthrough of 2013.
Bluestone’s UCSF lab was the first to show that CTLA-4 can turn off the immune system’s T-cell attack when studying its application to organ transplants and autoimmune disease, a discovery that help paved the way for Allison’s application to cancer.
He pointed to the University’s continuing role in pushing the field ahead, including recent work in cell therapy, which uses stem cells taken from a patient, corrects the gene mutation that causes disease and returns the “gene-corrected” cells to the patient for therapy.
This work is part of UCSF’s commitment to precision medicine, which aims to harness the wealth of genomic, clinical and environmental data from patients to develop more effective and even preventive therapies for human diseases.
“It has been a sea change that has been transformational,” Bluestone said, “not only from the patient’s point of view, but also for our understanding of the relationship between cancer and the immune system and how to treat disease.”
Immunotherapy’s unanswered questions
For all its success and promise, researchers still are baffled as to why immunotherapy works for some cancer patients, but not others.
“We have immunotherapy patients who live a long time, and now we can start to figure out why,” he added. “This is exactly what we’re focusing on in the lab now.”
In Fong’s lab, where he often works with Eric Small, M.D., deputy director of UCSF Helen Diller Family Comprehensive Cancer Center, his team is studying immune system activity in prostate cancer patients, trying to determine in which cases the immune response is activated and whether T-cells attack the cancer and why.
One of the most promising avenues is to combine immunotherapy treatments. Fong’s lab is now working on several studies regarding immunotherapy drugs, including a combination trial of sipueleucel-T and ipilimumab, which is scheduled to continue for the next couple of years.
For many aggressive, late-stage cancers, immunotherapy won’t replace traditional cancer treatments like chemotherapy, radiation and surgery, but it can be a complementary approach, said Lewis Lanier, Ph.D., chair of the UCSF Department of Microbiology and Immunology who collaborates with Fong on studying how cancer affects the immune system’s natural responses.
“Chemotherapy can reduce tumors by 95 percent, but if you have just one cell left, it can come back. So you can use [chemotherapy] to buy time, to really shrink the tumor if it’s far advanced, and then use the autoimmune drugs,” Lanier said.